Vinegar vs Baking Soda: When to Use Each
They sit in almost every kitchen. They’re cheap, safe, and endlessly recommended for cleaning. But most people use them interchangeably—spraying vinegar where baking soda would work better, or mixing them together and assuming the dramatic fizzing means powerful cleaning.
It doesn’t.
Understanding the actual chemistry behind these two ingredients transforms how effectively you clean. This isn’t complicated science—it’s knowing which tool solves which problem.
The Basic Science (Simply Explained)
Vinegar is acidic. White vinegar has a pH around 2.4, putting it firmly in acid territory. Acids dissolve alkaline substances—specifically mineral deposits, hard water buildup, calcium, and lime scale. Acids also cut through certain types of grease and kill some bacteria.
Baking soda is alkaline. With a pH around 9, baking soda sits on the opposite end of the scale. Alkaline substances neutralize acids, dissolve organic matter, and provide gentle physical abrasion when used as a paste or powder.
Here’s the critical insight most people miss: when you mix vinegar and baking soda together, they neutralize each other. The fizzing reaction produces water, carbon dioxide, and sodium acetate—none of which clean particularly well.
The dramatic bubbling looks like powerful chemistry happening. In reality, you’ve just cancelled out both ingredients.
Use them separately, sequentially if needed, and each becomes genuinely effective.
When to Use Vinegar

Hard Water Deposits and Mineral Buildup
This is where vinegar outperforms virtually everything, including expensive commercial products.
Hard water leaves calcium and magnesium deposits on every surface water touches—faucets, showerheads, toilet bowls, kettle interiors, coffee maker components. These chalky white or brownish deposits resist scrubbing and standard cleaners because they’re alkaline minerals that only acid can dissolve.
Vinegar’s acidity breaks these deposits down chemically. You’re not scrubbing them off—you’re dissolving them.
Showerhead cleaning: Submerge in a bag of undiluted vinegar for 2 hours. Mineral deposits blocking the holes dissolve completely, restoring water pressure without any scrubbing.
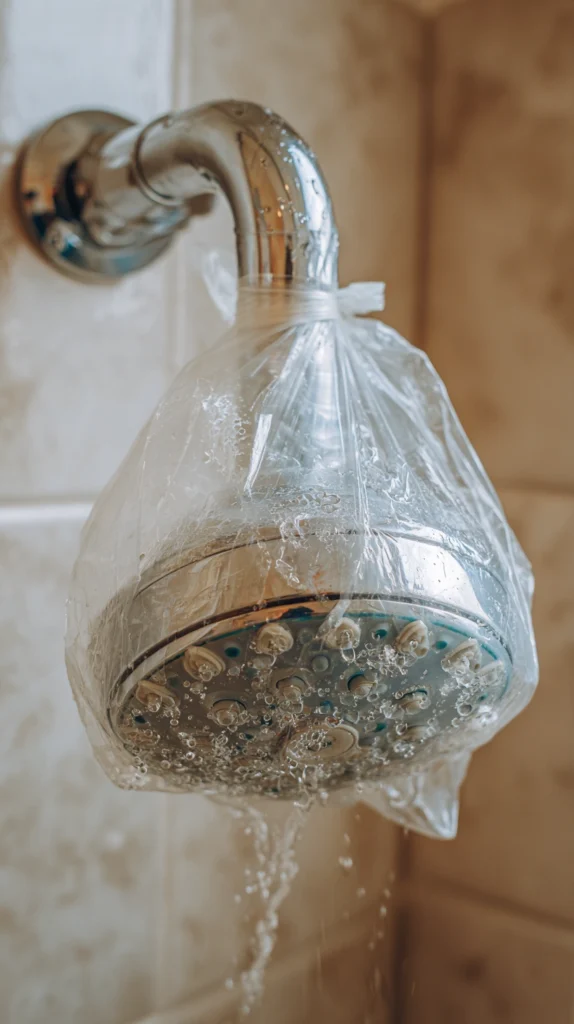
Kettle descaling: Fill halfway with equal parts vinegar and water, boil, let sit for 30 minutes, rinse thoroughly. Mineral buildup coating the heating element dissolves and flushes away.
Toilet bowl rings: Pour 2 cups vinegar into the bowl, let sit 2 hours or overnight for severe deposits. The mineral ring that resists scrubbing softens and wipes away.
Faucet buildup: Wrap fixtures with vinegar-soaked paper towels, leave for 30-60 minutes. The extended contact time dissolves what spray applications can’t.
Glass and Mirrors

Vinegar’s acidity cuts through the mineral film that makes glass look cloudy, leaving streak-free clarity that matches commercial glass cleaners.
Equal parts vinegar and water in a spray bottle, wiped with microfiber cloth. For stubborn water spots on shower glass, undiluted vinegar held against the surface for 5 minutes dissolves deposits that spraying can’t penetrate.
Odor Elimination
Vinegar neutralizes alkaline odors rather than masking them. It works specifically on odors with alkaline chemistry—pet accidents, musty smells, certain food odors.
Leave a bowl of white vinegar in a smelly room overnight. The smell dissipates as vinegar absorbs and neutralizes the odor molecules. The vinegar smell itself disappears as it dries.
For laundry, half a cup in the rinse cycle removes detergent residue and neutralizes odors from workout clothes and towels more effectively than fabric softener.
Coffee Makers and Kettles
Monthly descaling with vinegar solution prevents the mineral buildup that coats heating elements and eventually burns them out. This is one of the highest-value maintenance tasks for kitchen appliances.
Equal parts vinegar and water run through the machine, followed by two plain water cycles to rinse completely.
Fabric Softening
Half a cup of white vinegar in the washing machine’s fabric softener compartment softens clothes naturally by dissolving alkaline detergent residue left in fibers.
Clothes come out softer, towels stay more absorbent, and no chemical coating builds up over repeated washes.
Where Vinegar Fails
Natural stone surfaces: Granite, marble, slate, and other natural stones react with acid, causing etching and permanent dullness. Even diluted vinegar damages stone surfaces over time. Use pH-neutral dish soap instead.
Cast iron: Acid strips the seasoning that makes cast iron non-stick and rust-resistant. Never use vinegar on cast iron.
Waxed or unsealed wood: Vinegar strips wax finishes and can damage unsealed wood. Use very diluted solutions sparingly on sealed hardwood only.
Organic stains: Blood, wine, coffee, and food stains aren’t mineral-based. Acid doesn’t dissolve them effectively. Baking soda or hydrogen peroxide handles these better.
When to Use Baking Soda
Gentle Scrubbing Without Scratching
Baking soda’s crystalline structure provides mild abrasion that removes stuck-on residue without scratching most surfaces. This makes it ideal for situations where you need physical cleaning action without risking surface damage.
Sink basins, bathtubs, stovetops, and oven interiors all benefit from baking soda paste—enough abrasion to remove buildup, gentle enough not to scratch porcelain or enamel.
Mix with water to form a paste, apply to the surface, let sit for 10-15 minutes, scrub gently. The sitting time allows the alkaline chemistry to work before you add physical scrubbing.
Organic Stains and Food Residue

Baking soda’s alkalinity breaks down organic matter—food residue, grease, biological stains—more effectively than vinegar.
For baked-on oven residue, apply baking soda paste to the entire oven interior, let sit overnight, wipe clean in the morning. The extended contact time breaks down carbonized food residue that requires aggressive scrubbing without treatment.
For coffee and tea stains in mugs, baking soda paste scrubs away tannin staining without scratching the ceramic.
Odor Absorption
While vinegar neutralizes specific alkaline odors, baking soda absorbs a broader range of odors through a different mechanism—it chemically neutralizes both acidic and alkaline odor molecules.
An open box in the refrigerator absorbs food odors continuously. Sprinkled on carpets before vacuuming, it pulls musty and pet odors from fibers. Left in shoes overnight, it neutralizes bacterial odors.
Baking soda absorbs rather than masks—it doesn’t add a scent, it removes the existing one.
Laundry Boosting
Half a cup added to laundry along with regular detergent softens hard water, boosts cleaning power, and neutralizes odors in fabrics.
Particularly effective for gym clothes and towels that develop persistent smells despite regular washing. The alkalinity breaks down the bacterial compounds responsible for workout odors.
Drain Maintenance
Baking soda followed sequentially by vinegar provides drain maintenance cleaning—the baking soda loosens organic buildup, the vinegar fizzes through it, hot water flushes everything away.
Half a cup baking soda into the drain, followed after 5 minutes by 1 cup vinegar, followed after 15 minutes by hot water. This maintenance treatment prevents minor clogs and reduces odors from accumulated organic matter.
Note: this doesn’t clear actual clogs—it maintains drains between clogs.
Grease on Stovetops and Ovens

Baking soda is alkaline, and grease responds better to alkaline cleaners than acidic ones. Mix baking soda with a few drops of dish soap for maximum grease-cutting power.
Apply to greasy stovetop surfaces, let sit for 15 minutes, scrub with a damp cloth. For seriously baked-on grease, the overnight paste treatment works better than any spray product.
Where Baking Soda Fails
Mineral deposits: Baking soda is alkaline—the same chemistry as the minerals it’s trying to remove. It provides abrasion but no chemical dissolution. For hard water rings and lime scale, only acid works.
Disinfection: Baking soda doesn’t kill bacteria effectively. For sanitizing food surfaces or areas requiring genuine disinfection, use hydrogen peroxide or appropriate disinfectants.
Glass and mirrors: Baking soda leaves a white residue that’s difficult to rinse completely from glass surfaces, causing streaking. Vinegar handles glass consistently better.
The Sequential Approach
For certain cleaning tasks, using both in sequence—not mixed together—delivers better results than either alone.
Drain maintenance: Baking soda first to coat and loosen organic buildup, then vinegar to fizz through the loosened material, then hot water to flush.
Shower and tub: Spray vinegar to soften mineral deposits and soap scum, then apply baking soda paste over it and scrub. The vinegar pre-treatment makes the baking soda scrubbing more effective.
Oven cleaning: Baking soda paste overnight to break down organic residue, then vinegar spray to dissolve any remaining deposits and create a fizzing reaction that lifts loosened material.
The key distinction from mixing: you’re letting each ingredient work separately and sequentially, not combining them into a neutralized mixture.
Quick Reference Guide
| Cleaning Problem | Use Vinegar | Use Baking Soda |
| Hard water deposits | ✓ | ✗ |
| Mineral buildup | ✓ | ✗ |
| Glass and mirrors | ✓ | ✗ |
| Greasy residue | Moderate | ✓ |
| Organic food stains | ✗ | ✓ |
| Gentle scrubbing | ✗ | ✓ |
| Odor neutralizing | ✓ | ✓ |
| Laundry softening | ✓ | ✓ |
| Drain maintenance | Sequential | Sequential |
| Disinfection | Partial | ✗ |
| Natural stone | ✗ Never | ✓ |
| Cast iron | ✗ Never | ✓ |
The Practical Summary
Match the ingredient to the problem’s chemistry.
Seeing white chalky deposits, cloudy glass, or mineral buildup? That’s alkaline mineral chemistry requiring acidic vinegar to dissolve it.
Dealing with organic stains, food residue, grease, or needing gentle scrubbing? That’s where baking soda’s alkalinity and abrasion work best.
For odors and laundry, either works well depending on the specific situation.
Stop mixing them together expecting powerful results. Use them separately, where their individual chemistry actually solves the problem in front of you. Two inexpensive pantry staples become genuinely effective cleaning tools when applied correctly—and largely unnecessary when used wrong.






